Introduction: Convalescent plasma is a promising therapeutic option for corona virus disease 2019 (COVID-19). A recent study in 34 COVID-19 patients showed a reduction of recovered patients antibodies within 3 months of infection. The aim on this analysis was to evaluate the antibody titers and explore possible correlations with disease characteristics in volunteer donors, who participated in a phase 2 study for the use of convalescent plasma for the treatment of severe COVID-19 infection.
Patients and Methods: This in an ongoing phase 2 study (NCT04408209) for the use of convalescent plasma for severe COVID-19. This analysis reports the results of the first part of the study, regarding the presence of anti-SARS-CoV-2 antibodies in volunteer plasma donors and their correlation with disease characteristics. The main Inclusion criteria for plasma donors included: (i) confirmed SARS-CoV-2 infection by PCR of the nasal/pharyngeal swab; (ii) interval of at least 14 days after complete recovery from COVID-19; (iii) presence of anti-SARS-CoV-2 antibodies; (iv) two negative SARS-CoV-2 PCR results (the second at least 7 days prior to plasmapheresis). For the detection of anti-SARS-CoV-2 antibodies we used two commercially developed assays: one ELISA assay (Euroimmun Medizinische Labordiagnostika AG, Lubeck, Germany), which detects antibodies against the recombinant Spike protein of the virus (S1 domain) and a multiplex assay (ProtATonce Ltd, Athens, Greece) based on the Luminex® xMAP™ technology that detects total antibodies (IgG/IgM/IgA) and individual antibody isotypes IgG, IgM and IgA against 3 SARS-CoV-2 antigens (S1, basic nucleocapsid (N) protein and receptor-binding domain (RBD).
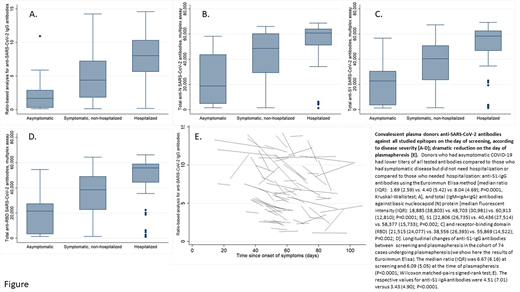
Results: To-date, 260 (137M/123F) possible plasma donors were tested for the presence of anti-SARS-CoV-2 antibodies. At the time of their COVID-19 diagnosis, 20 (7.7%) were asymptomatic, 157 (60.3%) were symptomatic but did not need hospitalization and 83 (32%) were hospitalized. Median time from the day of their first symptom or PCR+ (for asymptomatic patients) till the day of screening was 62 (range: 14-104) days. Anti-SARS-CoV-2 antibodies were detected in 229 (88%) donors with the Euroimmun assay and in 238 (91.5%) with the multiplex assay (including the 229 who had antibodies with the Euroimmun method). Univariate analysis showed that donors who had asymptomatic COVID-19 had lower antibody titer compared to those who had symptomatic disease but did not need hospitalization or those who hospitalized (Fig. A-D). Donors <50 years had lower antibody titer compared with older patients [for Euroimmun method, median (IQR): 3.94 (5.10) vs. 7.34 (6.16); p<0.0001], while patients who were tested within 60 days from the first day of symptom or PCR+ (for asymptomatic patients) had higher antibody titer [6.09 (6.52) vs. 4.68 (6.12); p=0.024]. The multivariate analysis showed that age ≥50 years (OR 2.88, 95% CI:1.60-5.18; p<0.001) and need for hospitalization (OR 4.11, 95% CI:2.13-7.90; p<0.001) correlated with higher antibody titers, while asymptomatic phase (OR 0.10, 95% CI:0,01-0.82; p<0.001) and testing within ≥60 days post symptoms onset (OR 0.36, 95% CI:0.20-0.66; p=0.001) correlated with lower antibody titers. In the multivariate logistic regression analysis examining associations between individual symptoms and antibody levels, there was strong correlation between anti-SARS-CoV-2 antibodies and anosmia (OR 11.14, 95% CI:3.92-31.67; p<0.001), loss of taste (OR 5.50, 95% CI:2.23-13.56; p<0.001), fever (OR 4.25, 95% CI:1.90-9.51; p<0.001), and headache (OR 2.34, 95% CI:1.09-5.03; p=0.029). To-date, plasmapheresis was performed in 74 patients with anti-SARS-CoV-2 antibodies, within a median time of 12 (8-19) days after screening; the respective median time (range) from the first day of symptoms or PCR+ was 52 (14-84) days. Interestingly, there was a significant reduction in the antibody titers between the day of screening and the day of plasmapheresis [Fig. E].
Conclusion: Lower anti-SARS-CoV-2 antibody titers, against all studied epitopes, are found in asymptomatic patients, in patients <50 years and in those who were tested ≥60 days post onset of first symptoms. The rapid reduction of anti-SARS-CoV-2 antibodies in our cohort reveals a time pattern of reduction, although we do not know if neutralizing antibodies share the same trend or if this reduction affects the host immunity against SARS-CoV-2.
Terpos:Amgen: Honoraria, Research Funding; BMS: Honoraria; Genesis pharma SA: Honoraria, Other: travel expenses , Research Funding; Janssen: Honoraria, Research Funding; Takeda: Honoraria, Other: travel expenses , Research Funding; Celgene: Honoraria; Sanofi: Honoraria. Pappa:Genesis pharma SA: Research Funding. Dimopoulos:BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Personal fees; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Personal fees, Research Funding, Speakers Bureau; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Personal fees, Research Funding, Speakers Bureau; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Personal fees, Research Funding, Speakers Bureau; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Personal fees, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal